Magnesium Oxide Nanomaterial For Carbon Dioxide Capture

KEY INFORMATION
TECHNOLOGY OVERVIEW
Pre-combustion, post-combustion and oxyfuel combustion capturing from power plants and other industrial scale companies are the three current carbon dioxide (CO2) capture and separation technologies. Unlike liquid and membrane adsorbents, solid adsorbents have a wider temperature range of adsorption and can be safely disposed in the environment. The use of solid adsorbents in industrial exhaust gases has shown to be a successful method of trapping concentrated CO2 for later storage rather than direct emission to the environment. Recent investigations have identified magnesium oxide based (MgO) solid adsorbents as a potential material for CO2 capture at intermediate temperatures. Furthermore, magnesium (Mg) based minerals are nontoxic, abundant materials which can be prepared in large scale at relatively low cost. Even though MgO has a high theoretical CO2 capture capacity (1100 mg CO2/g sorbent), it underperforms in practical applications due to a limiting number of active CO2 capture sites. MgO reacts with CO2 to create MgCO3 in dry, high-temperature circumstances. The formation of such MgCO3 carbonates obstructs additional carbon lattice transit leads which lowers the total CO2 capture efficiency.
This technology offer is an anion doping method of MgO at room temperature to prevent the formation of MgCO3. The novel MgO-Mg(OH)2 composite nanomaterial is formed via electrospinning technology and improves the overall efficiency of MgO as a CO2 capture material.
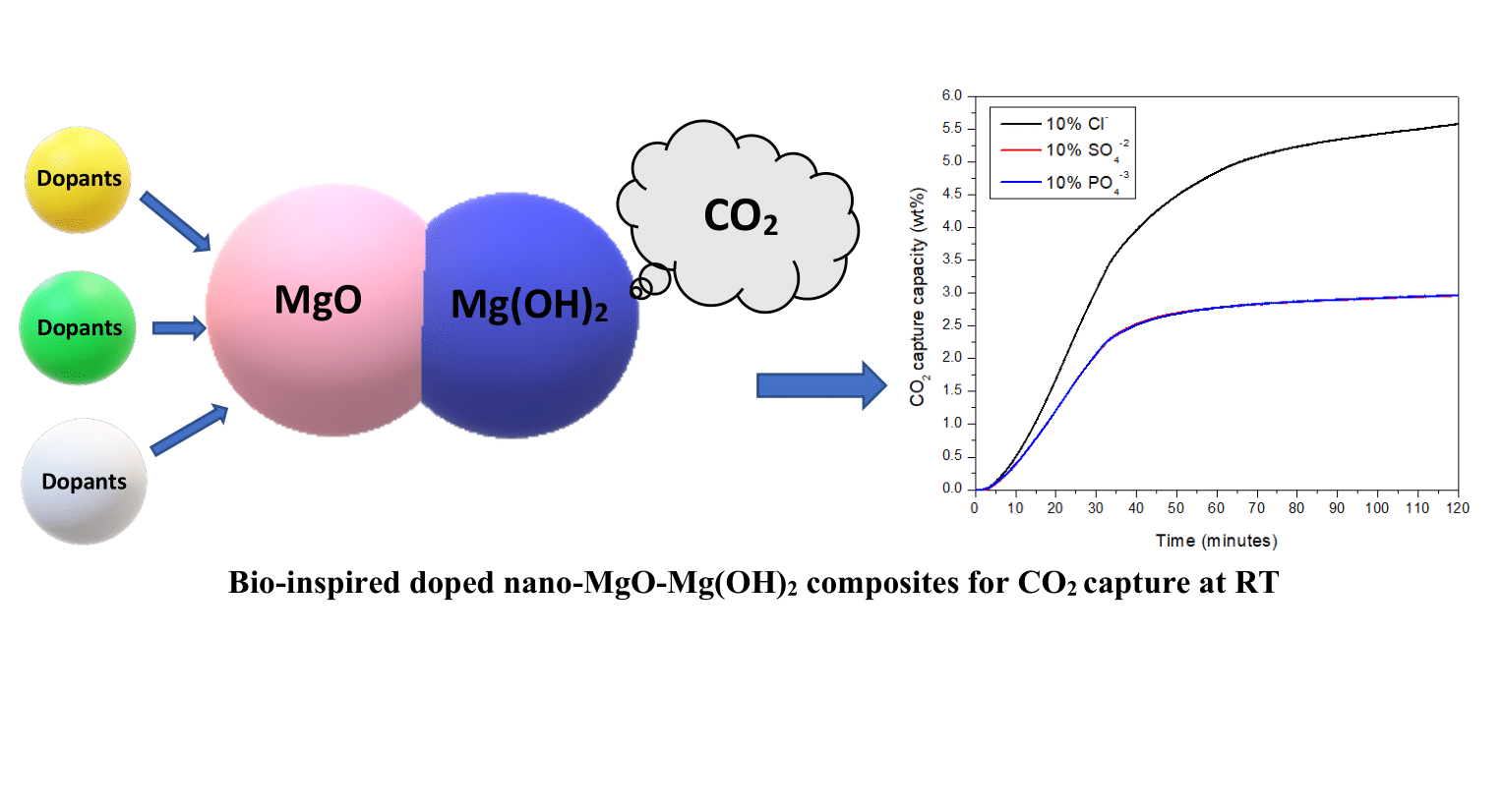
TECHNOLOGY FEATURES & SPECIFICATIONS
The doping was carried out by electrospinning technology in accordance with thermodynamic and quantum mechanical principles to improve process temperature and dopant/H2O concentrations in MgO-H2O-MgX (X= 2Cl-, SO42-, and 2/3PO43-) ternary systems. These novel composites aim to prevent the formation of MgCO3 to unblock the bulk diffusion of CO2 on MgO sorbents at 30 ℃ under 1 atm, by using anion anion-doped CO2-philic MgO and CO2-phobic Mg(OH)2. This technology can therefore be used as a room temperature CO2 adsorbents for applications such as indoor CO2 monitoring sensors.
POTENTIAL APPLICATIONS
This technology can be used for the following applications.
- CO2 monitoring sensors
- Room temperature direct air CO2 capture
- Industrial processes where large-scale carbon capture has been demonstrated
- Commercial operation including coal gasification, ethanol production, fertilizer production, natural gas processing, refinery hydrogen production and coal-fired power generation
Market Trends & Opportunities
Persistent atmospheric concentrations of greenhouse gases have now become a global issue, as they have a wide range of direct and indirect consequences on all living things on the planet. The most well-known result of this phenomenon is global warming, caused mainly by growing atmospheric CO2. CO2 is a major anthropogenic greenhouse gas, and the National Oceanic and Atmospheric Administration of the United States (NOAA) estimated that the average CO2 content in the atmosphere would be roughly 416.87 ppm at the end of December 2021, up from 338.80 ppm in 1980. As a result, scientists are actively developing solutions to minimize CO2 levels in the atmosphere.
The global carbon capture and storage market size was USD 2,784 million in 2021 and is estimated to grow at a CAGR of 13.7% from 2022 to 2030 and reach USD 8,636 million by 2030. The key markets drivers are:
- The surging investment to develop new capturing facilities
- The increase in government initiatives to achieve net-zero emission rates in the future
Unique Value Proposition
This technology addresses the limitation of MgO-based solid adsorbents and has the following advantages:
- Better carbon capture efficiency
- Cheaper than current CO2 adsorbent material
The technology owner is looking for partners for R&D collaborations especially those who are interested in carbon capture materials such as power plants or CO2 monitoring systems. The owner is also keen to license this technology as well.
